Compliance-first operations,from idea to approval.
OmniVynt is a compliance-first operations platform for pharmaceutical and life sciences companies, centralizing field activities, promotional content, and cross functional workflows in one system using the latest AI models.
Try it now
Enter a claim or use a sample to see our OmniVynt AI system flag risk in seconds.
Everything in one place.
Built for environments where claims, evidence, approvals, and budgets have to stay aligned—from first plan to final sign-off.
Compliance-first operations
Planning and collaboration designed specifically for regulated commercial and medical content.
AI-assisted design
Strategic project design, automated risk flagging, and intelligent claim rewriting.
Asset lifecycle management
End-to-end management of promotional content, documents, and media libraries.
Review & approval workflows
Structured, cross-functional collaboration and clear sign-offs across roles.
Budget governance
Complete budget visibility, tracking, and governance built directly into your workflow.
Cross-functional alignment
Connects reps, managers, medical affairs, and compliance teams in a single source of truth.
Three gaps we help close early
Fragmented workflows, compliance complexity, and weak claim discipline create delay. OmniVynt tightens alignment before formal review.
1. Fragmented Data & Omnichannel Coordination +
The Problem: Claims, evidence, drafts, and comments split across systems create version chaos and weak handoffs.
OmniVynt Solution: One system for claims, evidence, collaboration, and compliance decisions keeps work aligned before review.
2. Regulatory & AI Compliance Complexity +
The Problem: Teams are expected to move faster while proving every claim, output, and decision.
OmniVynt Solution: Source-verified flags and rewrite guidance strengthen submissions while keeping reviewer authority intact.
3. Asset Quality & Message Credibility +
The Problem: Weak claims, uneven fair balance, and lower asset quality reduce trust with HCPs, stakeholders, and patients.
OmniVynt Solution: Earlier claim-evidence and fair-balance checks strengthen Claim Credibility & Fair Balance while helping teams produce stronger review-ready assets.
From brief to submission-ready, in one workflow
Watch how OmniVynt surfaces claim risk, strengthens evidence alignment, and produces audit-ready outputs before formal PAAB, MLR, or other regulated review.
Advisory demo. Illustrative outputs only. Medical, legal, and regulatory teams retain final authority.
OmniVynt SaaS Feature List
A compliance-first planning, collaboration, content, and review platform built for commercial, marketing, medical, and review teams working in regulated environments.
Planning & Project Management
Build strategic project plans in a dedicated workspace. Track metadata, run AI-assisted drafting, manage files, and output printable proposals.

Content & Digital Asset Management
Manage your assets in a central hub. Upload with rich metadata, run version-based reviews, extract text from documents, and integrate with Adobe for supported workflows.

Compliance, Review & Risk Management
Threaded review histories, draft replies, and compliance visibility. Run AI compliance analysis reports on content assets and track issues across the platform.

Manager & Leadership Tools
Complete oversight of team activities, engagements, venues, and close-outs. Access budget controls, collaboration oversight, and comprehensive compliance tracking.

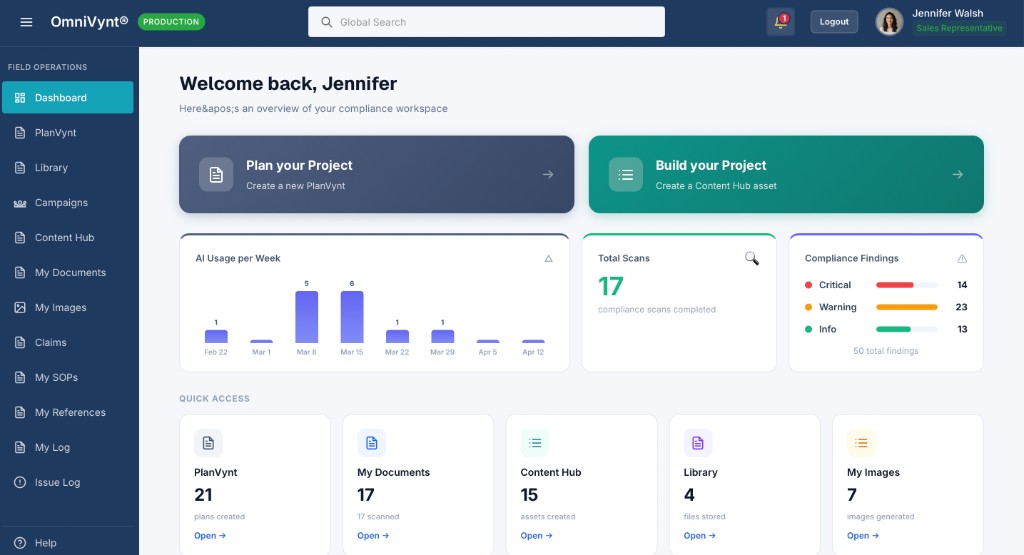
AI Image Generation & Library
Generate images for your content directly from prompts without worrying about copyrights. Organize your generated and uploaded assets into folders, edit metadata, and store them securely.
Core Platform
Secure login, role-based access for cross-functional teams, admin approval workflows, profile management, and a central dashboard with personal activity logs.
AI-Powered Strategy
Generate KPI, resource, and budget suggestions. Summarize project discussions with AI to develop structured strategy within a single workspace.
Collaboration & Chat
Dedicated project chat, threaded discussions, emoji reactions, email invitations, and direct file attachments in the collaboration feed.
Campaigns & Venues
Manage activities, venues, and close-outs. Run risk workflows on every activity, track objectives, attendees, and meeting types.
Document Library
Organizational folder sharing, extract text and summarize documents. Support for PDFs, Word, spreadsheets, and presentation files.
Image Generation
Generate images from prompts, upload your own, and organize into folders with detailed tracking of AI vs uploaded source labels.
Budget Oversight
Track allocations, spend vs allocation, global caps, and activity budgets. Access budget audit logs and policy settings.
Medical Affairs Workspace
Dedicated medical dashboard for scientific materials, medical information, scientific engagement support, HCP oversight, and SOP governance.
Operational Productivity
My Documents, cross-plan activity logs, search, filtering, status badges, shared UI patterns, and downloadable reports.
Why Specialty Brands Drift Earlier
Oncology, hematology, and rare disease teams operate with tighter claim boundaries, denser evidence packages, narrower patient populations, and less tolerance for drift. That makes upstream discipline more valuable and upstream mistakes more expensive.
Narrow Claim Boundaries
Small wording changes can push beyond approved population, line of therapy, biomarker context, intended use, or supporting clinical nuance.
Evidence Complexity
Claims often depend on precise endpoint language, subgroup context, comparator discipline, and stronger source linkage than general-market brands require.
Higher Rewrite Cost
By the time issues surface in review, teams may already have invested in creative, internal rounds, medical input, and launch timing that are expensive to unwind.
More Reviewer Scrutiny
Medical, legal, and regulatory teams spend more time where claims are dense, evidence is nuanced, and wording discipline matters more.
Concerns Are Team Specific
Different teams feel the same breakdown in different ways.
Reduce Rework and Protect Margins
Agencies feel the pain first through lost time, rework, and margin leakage. OmniVynt helps expose and reduce that hidden drag.
- Preventable revision rounds and rewrite work
- Version confusion across internal, client, and review teams
- Need for a credible commercial case before annual SaaS commitment
Enhance Launch Readiness and Reduce Risk
Brand teams feel pressure when claims, evidence, and approvals drift. OmniVynt strengthens work upstream so assets enter review cleaner.
- Launch delays caused by upstream drift
- More channels, stakeholders, and asset versions to manage
- Need for a cleaner handoff into MLR, PRC, or PAAB
Improve Review Efficiency with Full Human Oversight
Review teams need to see what was flagged, why it matters, and which evidence supports it. OmniVynt improves consistency while keeping final authority intact.
- No black-box recommendations
- Need cited, reviewable rationale
- Human authority remains non-negotiable
Tighter Claims, Denser Evidence, Less Room for Drift
In oncology, hematology, and rare disease, the cost of upstream errors is higher. Narrower indications, more precise endpoint language, and denser evidence packages mean wording discipline matters earlier — not just at formal review.
- Claim drift beyond approved population, line of therapy, or biomarker context
- Evidence linkage gaps that only surface late in the review cycle
- Higher rewrite cost when issues are caught at MLR or PAAB
The Real Cost Starts Before Review
Teams lose time before submission when claims come from multiple places, evidence is checked late, and versions multiply.
By then, the work is harder to defend, slower to fix, and more expensive to move into review.
OmniVynt Reduces Costs and Increases Efficiency Before Review
OmniVynt reduces risk and increases efficiency by catching common drafting issues before they create rework, delay, and review friction.
Three Issues OmniVynt Catches Early
Label Boundary Drift
Claims move beyond approved indications, intended use, or cleared parameters.
~25% of preventable resubmissionsClaim–Evidence Mismatch
Claims lack strong evidence or the right clinical framing.
~25% of preventable resubmissionsCitation & Reference Gaps
Missing or misaligned sources weaken defensibility in review.
~20% of preventable resubmissionsA Clearer Workflow From Planning to Review
OmniVynt helps regulated life sciences teams plan content and activities, create in one shared workspace, identify compliance issues earlier, and move assets into formal review with clearer communication and sign-off.
Plan content, collateral, activities, events, submissions, and more before creation begins
PlanVynt is the planning workspace for regulated marketing work. It helps teams organize content, collateral, activities, events, submissions, proposals, and other deliverables before creation begins, with clearer direction, shared communication, supporting material, and documented decisions in one place.
- Plan content, collateral, activities, events, submissions, and more
- Organize supporting material and source references
- Align teams on scope, direction, and responsibilities
- Keep early communication and decisions in one place
Create, review, and communicate in one shared workspace
Content Hub brings drafting, comments, revisions, version control, and review context into one place so teams stop losing time across email, calls, meetings, and disconnected files. It helps teams communicate more clearly, collaborate faster, and keep work moving without losing the thread.
- Create and review in one shared workspace
- Keep comments, revisions, and context in one thread
- Reduce communication drag across teams and partners
- Maintain version control across content and deliverables
Flag support, wording, and risk issues before formal review
Compliance Intelligence helps teams identify support gaps, risky wording, and potential compliance concerns earlier so fewer problems surface late in the process. It gives teams a cleaner path to review readiness by helping them strengthen work before it reaches formal review.
- Surface support gaps and wording concerns earlier
- Flag potential risk before formal review
- Improve alignment between content and supporting material
- Reduce preventable review pushback and rewrite cycles
Move cleaner assets forward with clearer sign-off
Review-Ready Handoff helps teams move assets into formal review with clearer approvals, attributed decisions, timestamps, and version history. That makes sign-off easier to trace, easier to defend, and easier to manage across multiple contributors.
- Keep sign-off clear and attributable
- Track decisions, timestamps, and version history
- Improve handoff into formal review
- Strengthen accountability across teams
Less Rework. Faster Launches. Protected Margins.
OmniVynt helps teams identify issues earlier, reduce rework, and move review-ready work forward faster.
Half the Time, Same Team
Asset development drops from 22.75 to 10.75 hours. Same headcount, roughly double the throughput — without cutting corners on compliance.
Fewer Rounds, Cleaner Submissions
Catch the flags that cause resubmissions before review begins. First-pass quality improves because risk is surfaced in week one, not at PAAB.
Compress the Launch Cycle
25–54 day internal cycles compress to 15–28 days. OmniVynt reduces the rounds your team controls, not PAAB’s process. Every internal stage gets faster.
Catch 70% of Preventable Issues
Label boundary drift, claim–evidence mismatch, and citation gaps account for the majority of preventable resubmissions. OmniVynt flags these before they reach formal review.
Build Your Business Case in 30 Seconds
Model how much time, rework, and operating value OmniVynt could recover for your team.
Model the Business Case
30 seconds. Scenario model only.
Prefer to walk through a modeled business case? Book Demo.
Four Ways to Work With OmniVynt
Start where it makes sense for your team. Every option is independent, no required sequence, no lock-in.
Not sure where to begin? A quick first signal: try the free demo scan. Ready to test on real assets: the 14-Day Proof Sprint is the most common first step. Specialty agency or validator: start with a pilot fit discussion.
- Enter any claim in the demo above
- See risk level, source flag, and rationale
- Glass-box output — no black box
- No sign-up required
- Full asset compliance scan
- Glass-box findings report
- Source-cited flags + rewrite options
- No platform commitment required
- Prioritized risk report across all assets
- Evidence-based rewrite options
- Documented business case + audit-ready docs
- 60-minute debrief + action playbook
- Full campaign and portfolio review
- Configured to your PM, SOPs, and markets
- Train-the-Trainer + compliance playbook
- Security package + enterprise terms
Each option stands on its own. If you start with the 14-Day Sprint and choose to continue, the sprint fee is credited in full toward Starter, Core, or Enterprise. If the sprint does not document clear value, the fee is waived. No lock-in at any stage.